News
Simulations Show Lander Exhaust Could Cloud Studies of Lunar Ices
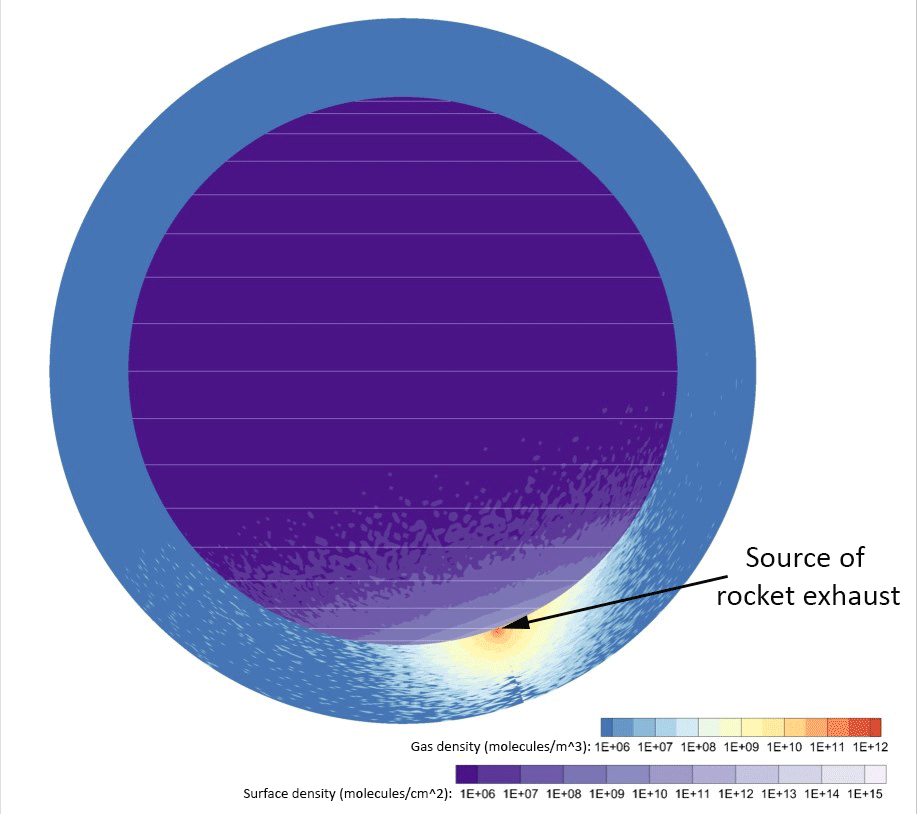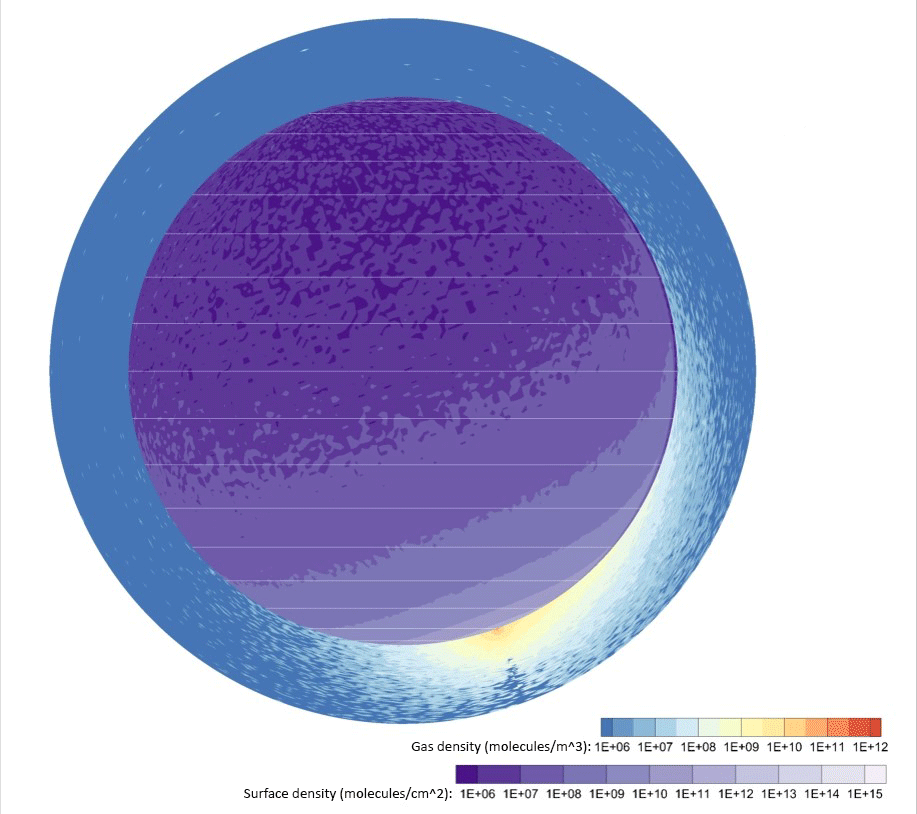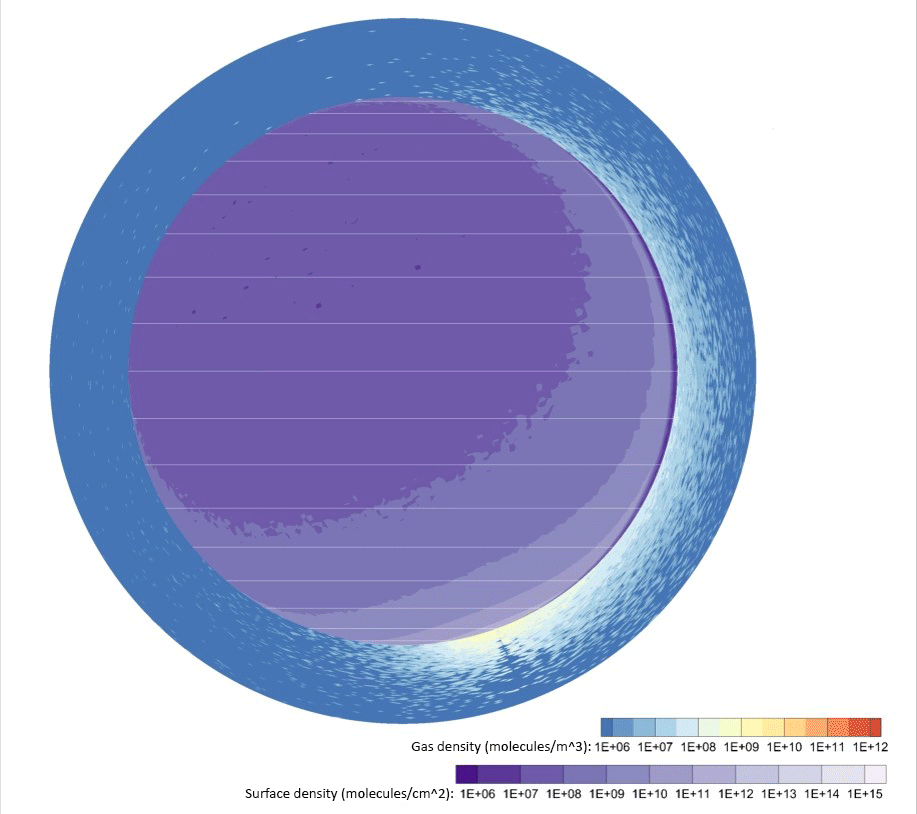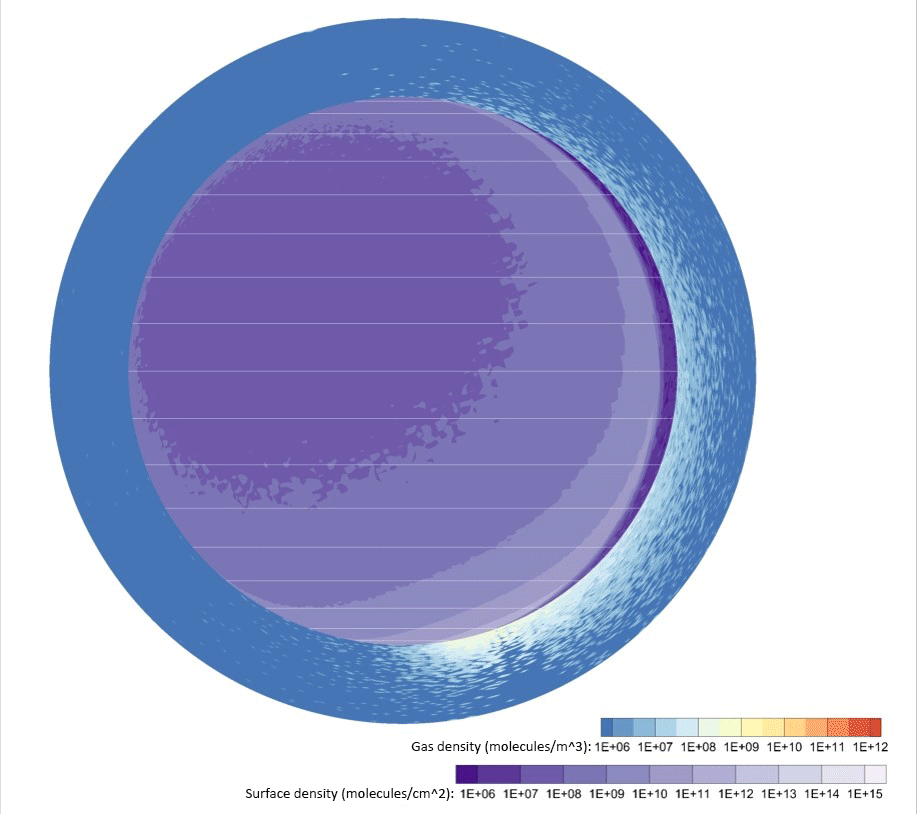
Credit: Johns Hopkins APL
A new study led by scientists at the Johns Hopkins Applied Physics Laboratory (APL) in Laurel, Maryland, shows that exhaust from a mid-sized lunar lander can quickly spread around the Moon and potentially contaminate scientifically vital ices at the lunar poles.
Computer simulations of water vapor emitted by a 2,650-pound (1,200-kilogram) lander — about a quarter of the dry mass of the Apollo Lunar Module — touching down near the Moon’s south pole showed exhaust takes only a few hours to disperse around the entire Moon. From 30% to 40% of the vapor persisted in the lunar atmosphere and surface two months later, and roughly 20% would ultimately freeze out near the poles a few months after that.
Those results, published online Aug. 11 in the Journal of Geophysical Research: Planets, show that researchers’ interest in studying the native ices in the Moon’s poleward craters — ices that may date back several billion years — will need to be carefully considered during increased efforts to return humans to the Moon.
Dealing with spacecraft exhaust on the Moon isn’t a new problem. Researchers appreciated this issue during NASA’s Apollo missions in the ‘60s and ‘70s, when they developed early models to predict the spread of exhaust throughout the lunar atmosphere and contamination of the surface.
“Exhaust during the Apollo mission didn’t complicate measurements in the same ways that it might now,” said Parvathy Prem, a researcher at APL and the lead author on the study.
During the Apollo era, most of the interest was in collecting lunar samples. While that’s still true today, the more recent discovery of ices preserved in permanently shadowed craters near the lunar poles has shifted scientific interest to understanding the origin and dispersion of water and other volatile molecules on the Moon’s surface and in its thin atmosphere.
“These are some of the only places where we can find traces of the origin of water in the inner solar system,” Prem said. Reading that record requires measuring the composition of those ices as well as their various isotopes to deduce where they likely came from and how they may have gotten there. Frozen-out exhaust gases from robotic or human exploration that collect on those ices could confound those measurements, even if the lander touches down hundreds of miles away.
“The interesting thing about Parvathy’s work is that it shows very well that the effect, while small and temporary, is global,” said Dana Hurley, a planetary scientist at APL and coauthor on the study.
Space organizations can expect volatile gases to significantly coat the lunar surface at well over 60 miles (100 kilometers) from the landing site.

Credit: NASA
The residue exhaust does eventually fade away, but Hurley points out that current plans for human lunar exploration mean it will happen more frequently and with much heavier landers.
“The results of this study drive the critical need to conduct the research we want to do about the lunar atmosphere and volatile deposits while they are relatively pristine,” Hurley said.
Prem cautions that the model isn’t foolproof. Among its most salient limitations are that it assumes the degree to which water interacts and “sticks” to the lunar surface, which is still uncertain but of great interest for understanding how easily water is transported around the Moon. The model also tracks only water vapor, which comprises about a third of the composition of most landers’ exhaust. Other exhaust molecules, such as hydrogen, ammonia and carbon monoxide, may behave differently and perhaps persist even longer.
Follow-up work should include measuring the amount of exhaust that’s around the Moon during and after future landings, Prem said, which would help narrow in on an answer to how much these exhaust gases “stick” to the surface. “But I would also suggest that modeling and monitoring the fate of exhaust gases should be a routine part of lunar mission development and planning.”
Conversations about mitigating exhaust gases have only just started, Prem explained.
In January, NASA finalized 16 science and technology demonstration payloads that it had selected to be delivered to the Moon through the Artemis program, including the Surface Exosphere Alterations by Landers (SEAL), an instrument that will investigate the chemical response of the lunar surface during a landing as well as any contaminants that may have been injected.
“Whether we intend to or not, we’re going to do this experiment of bringing exhaust gases with us,” Prem said. It’s now a matter of deciding how we deal with them.
